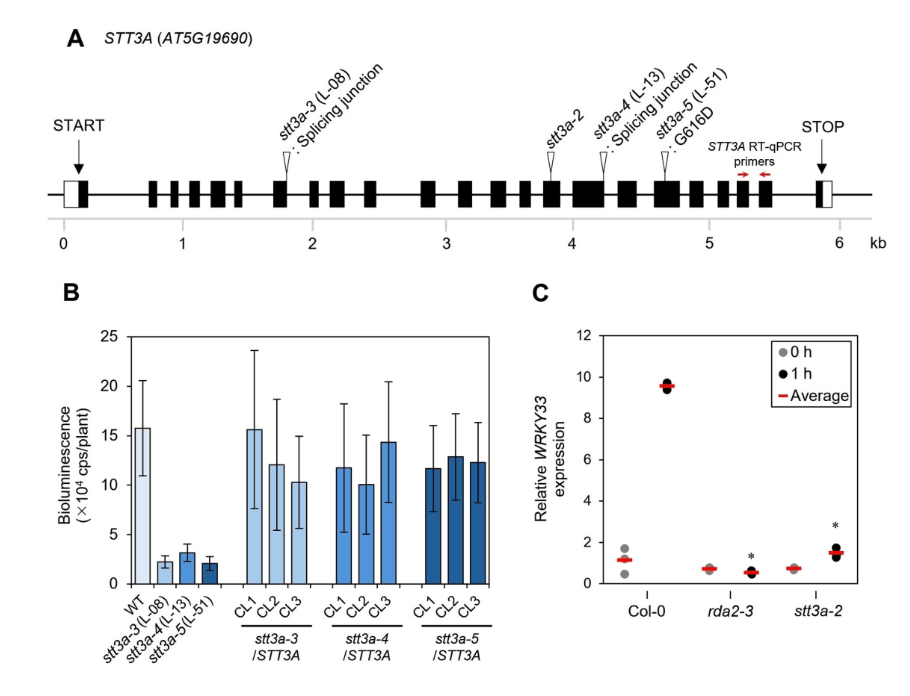
Abstract
Plants recognize pathogen-associated molecular patterns (PAMPs) via pattern recognition receptors, leading to the activation of pattern-triggered immunity in response to pathogen attack. Phytophthora infestans ceramide D (Pi-Cer D) is a sphingolipid from the oomycete pathogen P. infestans. Pi-Cer D is cleaved by the plant extracellular ceramidase NEUTRAL CERAMIDASE 2 (NCER2), and the resulting 9-methyl-branched sphingoid base is recognized by the plant receptor RESISTANT TO DFPM-INHIBITION OF ABSCISIC ACID SIGNALING 2 (RDA2) at the plasma membrane to transduce a defense signal. However, additional components are likely involved in sphingolipid recognition, which remain to be identified. Here, we employed a screen based on Lumi-Map technology to look for Arabidopsis (Arabidopsis thaliana) mutants with altered defense responses to Pi-Cer D. We identified three mutants showing diminished responses to Pi-Cer D and elf18, each carrying mutations in STAUROSPORIN AND TEMPERATURE SENSITIVE 3-LIKE A (STT3A), which encodes an oligosaccharyltransferase. The stt3a mutants exhibited higher susceptibility to the pathogen Colletotrichum higginsianum than the wild type. In stt3a mutants, the molecular mass of NCER2 and RDA2 proteins appeared smaller, indicating STT3A is involved in post-translational modification of the proteins. Enzymatic deglycosylation assay revealed that NCER2 and RDA2 are N-glycosylated. These findings suggest that STT3A contributes to plant immunity via post-translational modification of proteins including NCER2 and RDA2.